Overview
Enabling RNA therapeutics in linear, circular, or oligo formats
RNA therapeutics are programmable medicines. The efficacy, safety, and stability of an RNA drug is determined primarily by its sequence. Ginkgo’s foundry enables our partners to explore large genetic design spaces using both model-guided and unbiased methods. With the power of automation and foundry-scale datasets, we unlock novel functional RNA sequences optimized for your application.
The right RNA sequence needs the right vector and a scaled-up manufacturing process. Ginkgo offers end-to-end support for RNA therapeutics developers from early discovery to proof-of-concept animal studies. We are your discovery partner across all the formats in which you design and deliver RNA.
RNA formats
- mRNA
- Circular RNA
- Self-amplifying RNA
- Non-coding & oligomeric RNA
Solutions for
- RNA stability
- Immunogenicity
- Endosomal escape
- Strong & sustained expression
- Cell & tissue specificity
- Production & purification processes
Building the next generation of RNA vaccines and therapeutics? Lets circle up.
Our capabilities span the process of creating new RNA therapeutics from early discovery to sample preparation for animal studies. Partner with us at any stage for as much or as little work as you need.
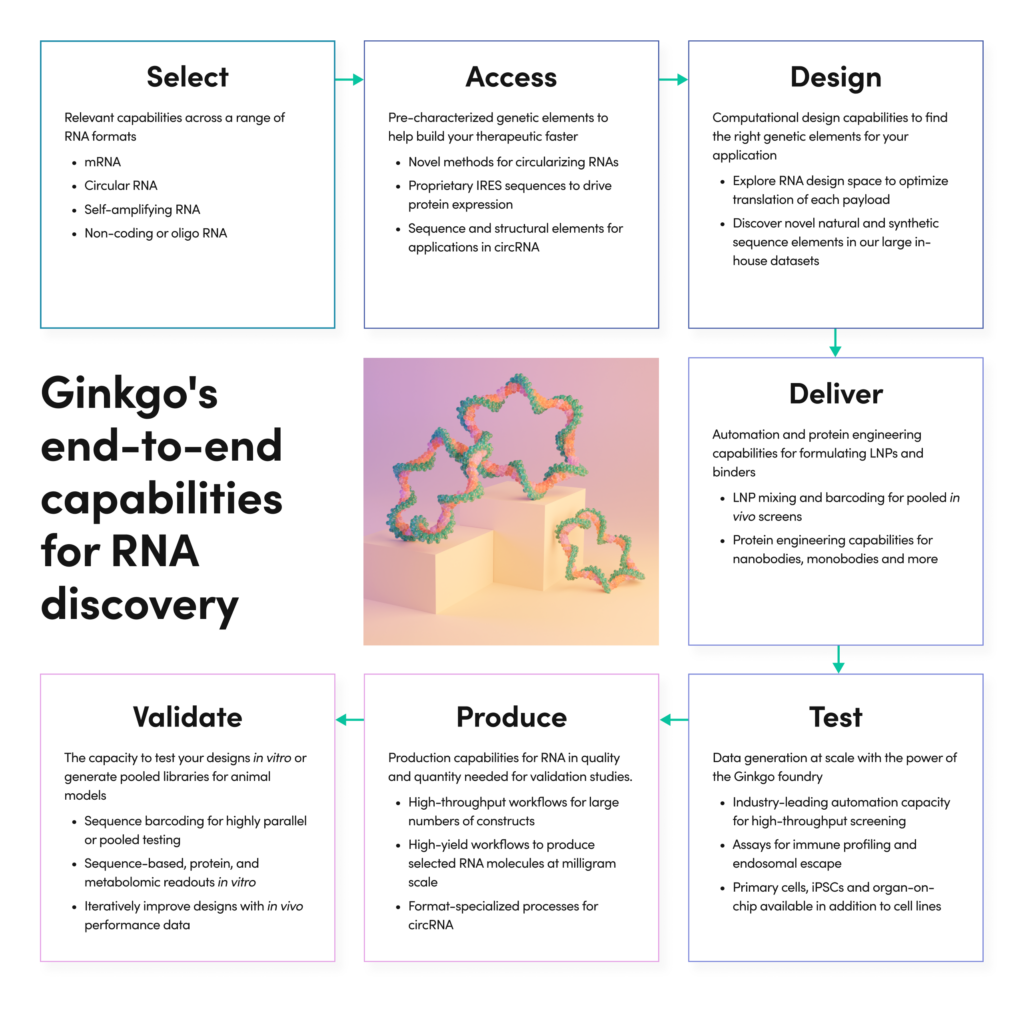
01 SEQUENCE DISCOVERY
Unlock novel and optimized RNA sequence elements
The versatile RNA molecule can deliver therapeutic activity in many forms: coding sequences, sequence-specific binding interactions, regulatory elements, aptameric or enzymatic activities. Ginkgo’s discovery services help our partners to identify, characterize and optimize the functional elements that make RNA work for therapeutics and vaccines.
At the heart of our discovery services is the foundry, an expansive facility for genetic design and high-throughput screening. The foundry brings scale to the discovery process, allowing us to explore large design spaces and compare performance in cell-based assays.
Discovery is data-driven. Ginkgo’s partners gain access to our large in-house datasets of characterized functional RNAs. Make use of existing functional elements that meet your performance needs, or design novel RNA constructs informed by our data resources.
Examples of RNA discovery projects enabled by the Ginkgo foundry
- Find IRES elements to drive tissue-specific expression from a circular RNA.
- Compare mRNA and circular RNA designs head-to-head.
- Design antibodies or other binding elements for targeting delivery.
- Optimize mRNA 5′ UTRs & 3′ polyA sequences.
- Discover novel introns or ribozymes to circularize your RNA.
- Control the expression strength of a protein payload.
- Engineer synthetic protein payloads with ML-guided design tools.
- Explore construct activity in vivo with barcoded and pooled screening.
02 CIRCULAR RNA
Improve stability and reduce immunogenicity with circular RNA
Circular RNA is inherently protected from endonuclease activity, giving it enhanced stability in many in vivo contexts. Because conventional mRNA end motifs like the 5′ cap and 3′ polyA tail are recognized by certain immune responses, circRNA may also be less immunogenic.
Working with RNA in the circular format means adopting new design elements, in particular the IRES (Internal Ribosome Entry Site) sequences that can initiate translation without a 5′ cap. Our partners benefit from our large library of pre-characterized sequence elements to streamline the design process. The right RNA sequence elements can deliver your therapeutic payload at the right dose and with cell and tissue specificity, improving efficacy and safety profiles.
Ginkgo leverages a variety of techniques to circularize RNA, including technology acquired from Circularis in 2022. We can explore existing methods systematically for performance in your application, or mine Ginkgo’s large in-house DNA sequence libraries for novel circularization elements.
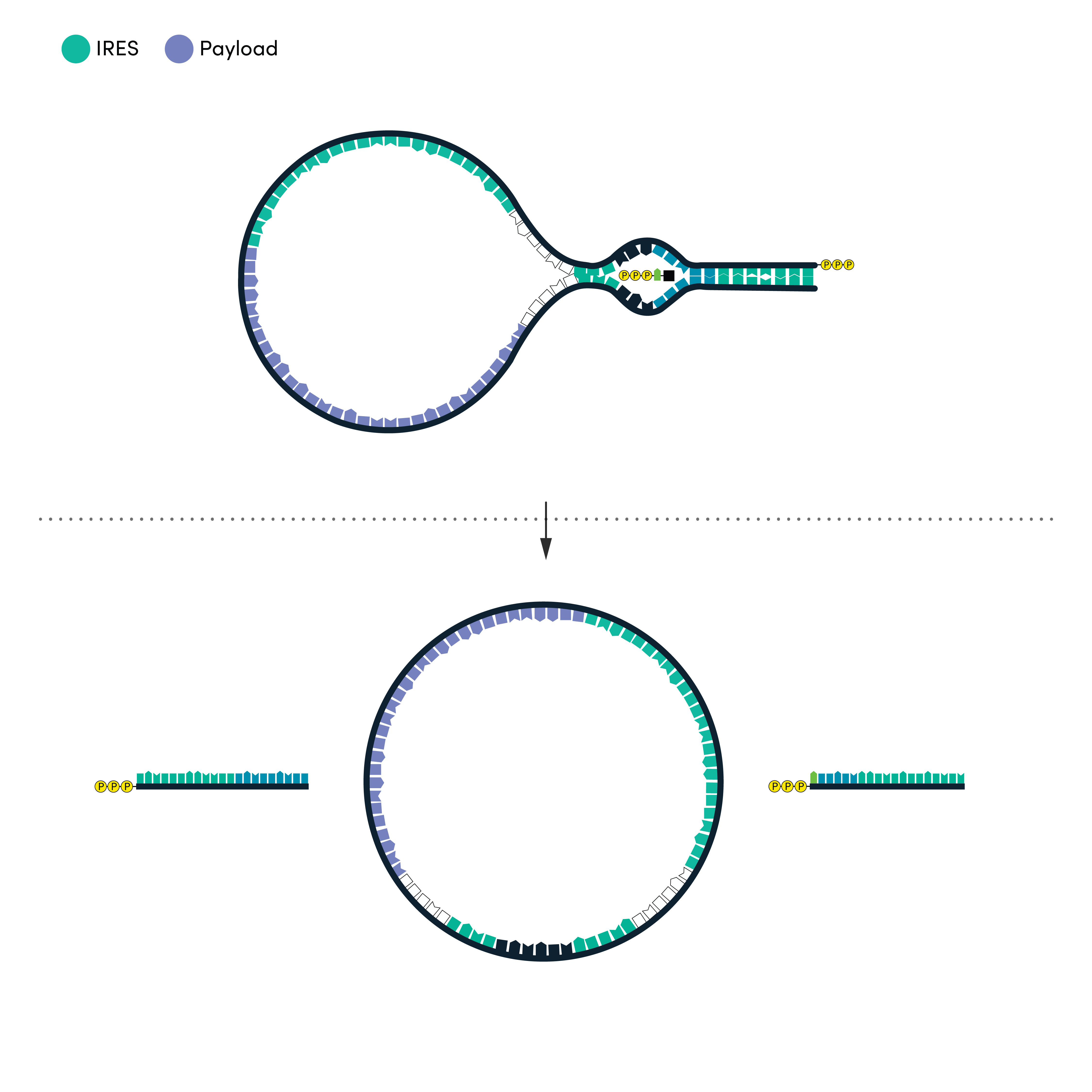
RNA circularization options
- Self-splicing by Group I intron
- Self-splicing by Hairpin ribozymes
- Enzyme-mediated RNA ligation
- Spliceosome-mediated
03 NON-CODING AND OLIGO RNA
Automated workflows for optimizing small RNA oligos
Even small RNAs or RNA-like therapeutics often need to explore a large sequence space before landing on the right design. Ginkgo has extensive capabilities in RNA oligo synthesis, including with modified bases. We can screen large libraries to optimize for selectivity, immunogenicity, or with custom assays for your therapeutic mechanism of action.
Small RNAs and RNA-like therapeutics in all shapes and sizes
- ASOs (Antisense Oligos)
- PMOs (Phosphorodiamidate morpholino oligomers)
- siRNA & saRNA
- lncRNA
High-throughput screening for
- Gene expression
- Immunogenicity
- Endosomal escape
04 PRODUCTION & PURIFICATION
Scaled-up RNA production in high yield or high throughput
A good RNA design can’t advance to the clinic unless it can also be produced with high titer and purity. At Ginkgo we can design RNA for manufacturability from day 1, incorporating sequence motifs that stabilize expression and support stability.
Because circRNA requires special care during production and purification, the services that we offer for RNA include specialized workflows in HPLC and FPLC to meet our partners’ production needs.
Ginkgo’s RNA production workflows regularly achieve 80-90% purity, depending on RNA sequence, size and the required scale of production. Our RNAs synthesis processes adhere strictly to biopharmaceutical industry standards across a range of RNA formats.
- High-throughput production of many different sequence variants at the microgram scale to support comparative and pooled screens
- High-yield production of single RNA molecules at the milligram scale for larger studies.
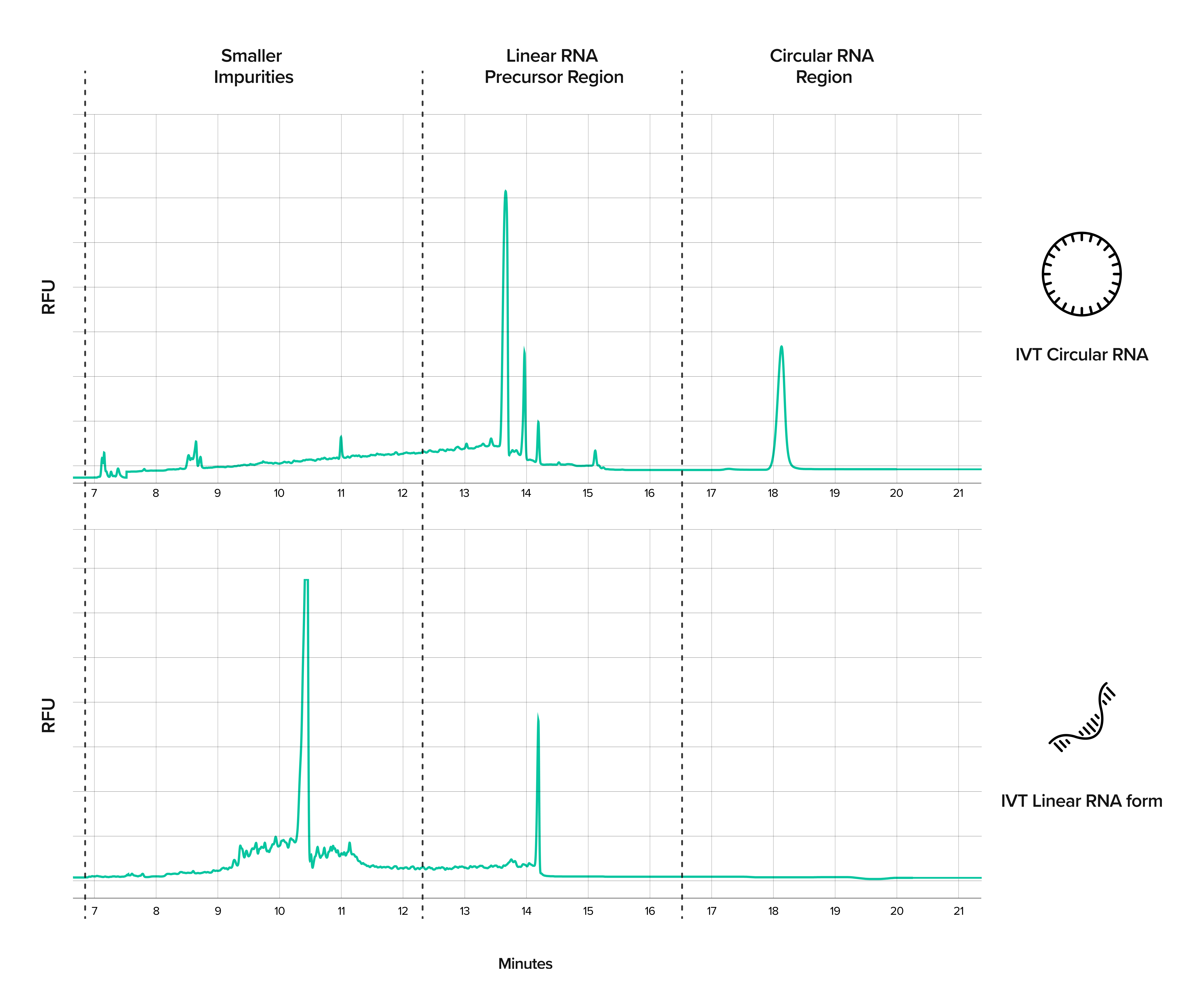
RNA production processes to support
- 5′ capping
- Modified bases
- Circular RNA
05 TARGETING & DELIVERY
Explore LNP formulations, develop binders for targeted delivery
LNPs (Lipid Nanoparticles) are versatile non-viral vectors to stabilize an RNA therapeutic and deliver it to a specific tissue or cell type. Finding the ideal LNP formulation for your application means generating large numbers of compositions and comparing their performance in vivo. Our high-throughput liquid handling capabilities can support our partners in exploring LNP formulations.
Even more cell-type specific targeting can be achieved with the use of engineered binders. A binder may be an antibody or another peptide with affinity for a specific tissue or cell type. Ginkgo has extensive capabilities in protein engineering and high-throughput screening to create binders with the desired affinity and selectivity.
Explore binders to target your RNA therapeutic
- ScFv
- IgG
- Fab
- Peptides
- Cyclic peptides
- …and more
Ultra-high-throughput binder engineering with EncapS
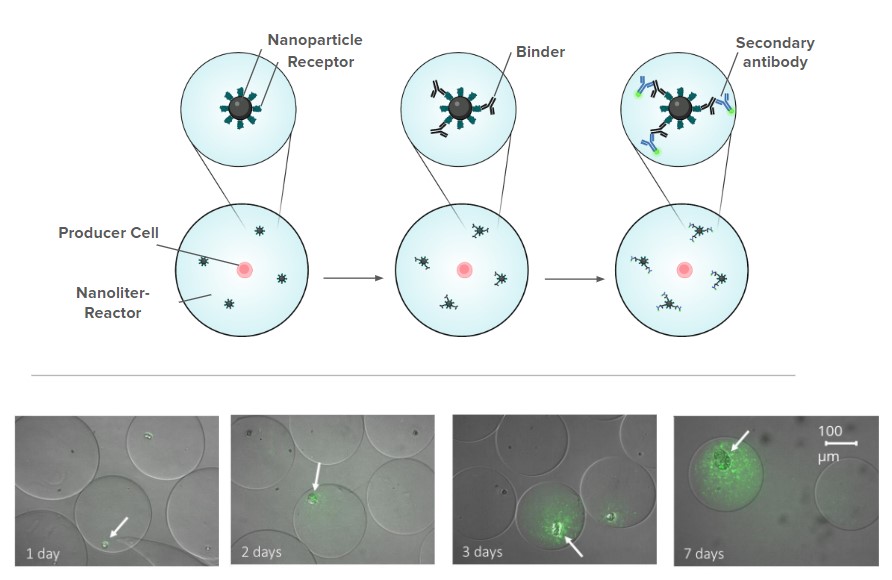
EncapS (Encapsulation & Screening) is Ginkgo’s proprietary technology for characterizing binder-producing strain libraries in nanolinter bioreactors. Individual cells grow into functional microcolonies, allowing screening for complex phenotypes.
EncapS for binder discovery
- Screen 100,000+ variant libraries in a single run.
- Optimize for affinity, titer and other functional properties
- Co-encapsulation enables selection for complex phenotypic responses
HOW WE PARTNER
An innovation partner with tech you can’t get anywhere else
Ginkgo Bioworks is a pure platform company. We’re dedicated to helping our partners to advance their RNA therapeutics rather than seeking to develop our own biopharma products in house. We offer a variety of partnership models to meet your technical needs, whatever your stage of the R&D process.
Our partners gain access to our massive genetic datasets, our library of characterized genetic elements for RNA and circRNA, and our industry-leading foundry automation infrastructure.
Why work with Ginkgo to develop your RNA therapeutic?
- Take control of your R&D spending by replacing fixed costs with variable costs.
- Avoid costs and delays required to build your own infrastructure.
- Launch faster by starting from pre-characterized genetic elements.
- Gain access to our world-class team of synthetic biology experts
"*" indicates required fields